Dry Film Thickness, wetting out, and subsurface corrosion.
Why do we break our own coatings?
Ever read an advert on someones product, and come away not believing a word of it? Product information likened to a political party manifesto?
We believe that every coating is a compromise. In an ideal word metals would not react, they would not degrade, corrode, rust, or have correlating reactions with other metals and substances.
So we cover these substrates in a range of products to seek to isolate them in some manner. We have Mono-molecular VCI coatings that use "just enough" and are limited to a narrow set of parameters where they flourish (such as an enclosed cavity with little air movement), right up to near 100% solids two pack epoxy, and hot dipping processes like galvanising. All of these processes seek to isolate the substrate from something else (typically atmosphere, but often includes submersion and other metals) and that doesn't always go right.
We have a huge range of contributing factors and we will cover that another day.
The focus of this blog post is to cover off some basics with destructive testing we have done with CIO specifically.
We have here a piece of steel withe three Different types of CIO.
1. TOP CIO Neat. not thinned.
2. MIDDLE CIO Thinned 50% using CIO thinners
3. BOTTOM CIO Thinned 50% using Tetrosyl thinners
The black vertical line represents the divide between the panel wiped side, and the non panel wiped side. As you can see the panel wipe does not fully evaporate. It offers a basic level of protection for the steel (left side has more steel that is yet to corrode). However, we can see it has no affect on the coating performance in this situation (steel was not contaminated prior to application).

Moving on from the above, we wanted to not only test the affect of panel wipe, and thinners, we want to look at DFT (Dry Film Thickness). DFT is the thickness of the coating once cured. Solvents will flash off during the curing process, and some degree of shrinkage will occur.
Applying a product to the correct DFT is critical. You are applying a layer ontop of the substrate and you want it to protect all of it. Looking under a microscope steel will look like this
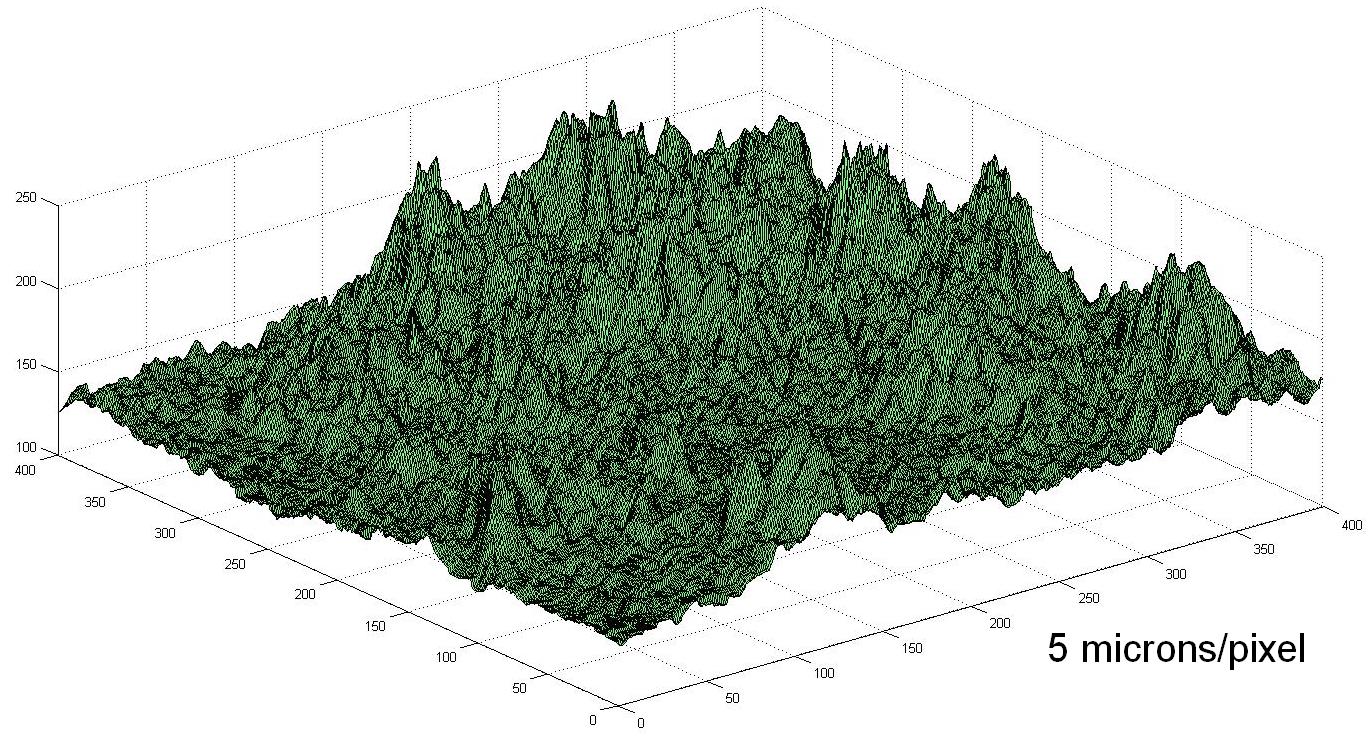
So we have Peaks and valleys that will in basic terms look like this.
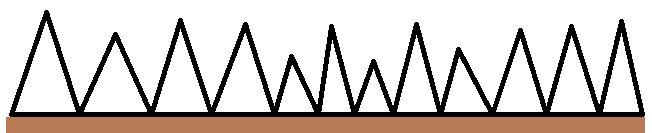
The wetting out properties of our coating will affect how well the valleys of the substrate are followed, and the build, or DFT ontop of that wetting out property will affect the total product above the substrate. For example a product that wets out well, but you don't put enough on will leave the peaks of the substrate exposed, leading to premature rust spotting.
The picture below demonstrates a coating that has wetted out perfectly (a near impossibility), but has insufficient DFT. This is usually caused by not enough product being applied. Such as spraying too fast, thinning too much, or spraying from a poor angle leading to only a light covering, or brushing out too far.
Points in diagram.
- Paint applied.
- Coating wetted out perfectly following the valleys of the substrate. This means that air and moisture are not trapped there, which will typically lead to blistering later on.
- Peaks of the substrate are either exposed, or not covered with sufficient DFT to allow sufficient protection. The result of this is spot rusting, and premature failure of the coating.
- Peaks not sufficiently covered will likely become exposed from abrasion, damage, and wear and tear of the coating. Additionally coatings become thinner, and/ or more porous as time passes. As a result these vulnerable peaks will become a contributing factor later on.
- Thinned down products tend to offer poor DFT. A common one is waxoil mixed with engine oil.

On our test sample you can see this. The test sample has been angled so that each test piece has a low and a high DFT area. All Low DFT areas are exhibiting signs of failure because the steel is literally poking through the surface of the paint, and has not been covered correctly. Additionally the cheaper thinners are causing significantly more issues because they are damaging the binder of the paint, and causing porosity and adhesion issues - A good tip is to ALWAYS use the correct thinner.

Next up we have a coating that is poorly made/ designed, or the preparation is insufficient (such as a polished surface), The result of this is that the paint sits ontop of the substrate and either fails to adhere and chips off, or traps moisture and gets blown off with sub surface corrosion (blistering) later on. Stonechip and thick underbody waxes over rust tend to do this.,
- Trapped moisture and air.
- External appearance is fine.
Bitumen based, schutz, and most underseals and stone chips do this to correctly prepared surfaces. They rarely wet out correctly, and look great when finished short term. Additionally because of their very thick coating (high dft) they mask the sub surface corrosion underneath. Wet waxes are also prone to this, which is why they should be applied to well prepared, dry rust free surfaces typically.

When thinning a product, you are reducing the hold of that product on corners and edges, and the film thickness, given that it will now flow and run easier. This can assist wetting out properties, but will leave peaks exposed. Peaks and valleys are present on all metals that we typically coat, and as a result of that we must ensure we pick the correct product for the preparation we are able and willing to do. There is no point selecting the hardest, thickest paint on the market, if you are not willing to give it the key, and substrate it requires.
Its not worth doing all the preparation anyone could ever hope for, if your going to use a product that won't suit your needs. Such as doing a nut and bolt rebuild, and using the cheapest paint you could possibly find, that will almost certainly be a low solids coating, full of filler, with terrible wetting out properties, and low DFT, almost certainly with a big sticker stating it contains "corrosion inhibitors".
Match the coating, to the preparation, and those to your expectations and your budget. Read more on CHOOSING A CHASSIS PAINT